About us
Goldshield® technology
OUR PHILOSOPHY
Trust Goldshield to Protect What Matters.
With the rise of “superbugs”, COVID 19 [SARS CoV2], MRSA, Staph, and mould spores, there has never been a stronger awareness in the public mind about infections and the devastation that a global pandemic can bring to bear on us all. Each organisation must now balance the health and safety of their employees, adhere to new and rapidly-changing government guidelines and provide peace of mind.
Goldshield is here to help and we can help you make a real impact with transparency, confidence and comfort safe in the knowledge that our global research and ground-breaking technology is available today and at your disposal. Combined with the powerful residual protection of Goldshield, you can be assured you’re providing the most effective, safe, and eco-friendly germ control system the world has to offer. Goldshield is the only technology to be peer-reviewed on 4 separate occasions and without doubt our patented technology is now widely regarded globally as being the preferred choice by those who know best.
Trust Goldshield
To Protect
What Matters

OUR STORY
In the middle of years of research on antimicrobial technology, our co-founder, Tom Higgins’ father passed away from a hospital-acquired infection. He found himself newly driven toward developing technologies that would eliminate the possibilities of germs, when improperly cleaned with ineffective, poor performing products, killing another person’s loved one.
With the help of his partner, Ted Shlisky and a former Dean of Harvard Medical School, in 2008 the Goldshield Formula was born and through a process of non-stop research and development of innovative residual technology for hands, surfaces, laundry, FFP3 face masks and many more besides. Goldshield today is proud to deliver the best protection possible and the family continues to work tirelessly towards his dream of spreading this safe protection to every corner of the world.

Original 8oz. GS24 Hand Sanitiser & 32oz. GS75 Surface Protectant
THE SCIENCE
Goldshield safely and quickly bonds to any surface or textile. It protects against everything from microbial contamination to drug resistant biologics, mould and mildew. This eliminates the need for exhaustive disinfectant re-applications and the use of substantial body protection when applying.

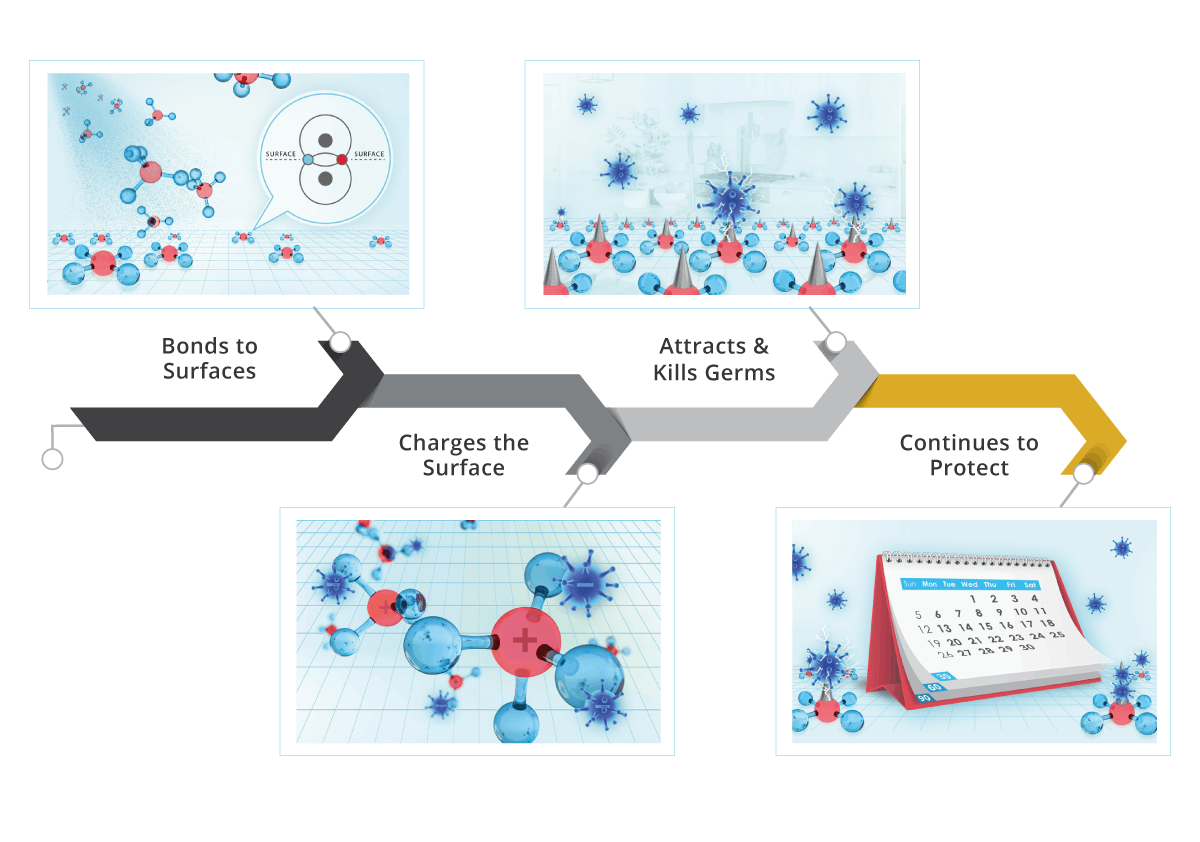
Goldshield has a nitrogen molecule. This nitrogen positively charges Goldshield – it’s called electrostatic charge. Most germs are negatively charged. So, like strong magnets, germs are “attracted” to anywhere Goldshield has been applied.
Each tiny spike can kill multiple germs depending on size (think germ kabab). It typically takes 90 days before all the spikes hit dead germ capacity. Because of Goldshield’s bonding ability, your surface and fabrics are protected for up to 90 days.

THE DIFFERENCE
The scientists at Goldshield have spent years perfecting the science of antimicrobial protection in a nano-assembly. The results of our proactive, residual performing technologies provide safer environments as well as time and cost savings.
Our products are the world’s only patented, water-stabilised antimicrobial formulation that breaks down surface tension and imparts a durable, antimicrobial protective shield to nearly all surfaces, textiles, and skin.
Safe to Use
- EN, EPA & FDA registered
- Water-based
- Approved for hospitals
- Alcohol, methanol, bleach and phosphate-free
- Non-flammable, combustible or corrosive
Long Lasting
- Protects between uses once applied depending on substrate and product
- Select products proven effective against COVID-19
- Up to 99.99% effective against other illness-causing germs, bacteria, fungi (Mould and mildew), algae and yeast on contact
Patented & Proven
- Privately held Patent# USA 9,089,138
- 65+ evidence based studies
- Approved for hospitals
- 4 key third-party, peer reviewed publications, including the American Journal of Infection Control
Non-Leaching
- Forms a non-migrating bond
- Won’t leach into the bloodstream
- Does not pose the risk of creating “Super-bugs”
OUR RELATIONSHIPS
GLOBAL APPROVALS
Our products adhere to the strictest guidelines outlined within these organisations.
- Goldshield 5 Antimicrobial Surface
Protectant (Concentrate)
Registration # 85556-1 - Goldshield 75 Antimicrobial Surface
Protectant (Ready-to-use)
Registration # 85556-2
- Goldshield 24 Antimicrobial & Antiseptic Moisturising Hand Sanitiser
NDC Code 40092-500-12
- Goldshield 5 Laundry Protector (Concentrate) EN 14476, EN 1650, EN 1656, EN 1647
- Goldshield 75 14 Day Surface Disinfectant & Protector (Ready-to-use) EN 14476
- Goldshield 24 24 Hour Hand Sanitiser EN 1656, EN 1657, EN 1276, EN 1500
- Goldshield GO36 FFP3 Mask CE-PC-210208-054-01-9A
Each region of the world has their own regulatory agency that tests products to prove their kill claims. Each agency has their own “testing protocols”. In the absence of a standard testing protocol, there is no way for a product to be approved for a claim. In the case of disinfection claims you are required to be tested for both bacterial and viral pathogens.
The United States had begun the process of developing a “residual viral” testing protocol prior to the pandemic in 2019, but due to the high demand, high stress bought on by the pandemic for the agency, that process was interrupted and has still not been completed as of September 2021. Until that is completed, in the United States our Goldshield Antimicrobial Surface Agent products are not permitted to use the word “disinfectant” or “virus(es)” on our labels or website.
NOTE: The EPA Homeland Security Laboratory and scientists have been using Goldshield to test the planned residual viral protocol for efficacy and we have received very favorable results. Unfortunately, the department of Efficacy at the EPA (those responsible for approving claims for labels) considers testing done in the EPA’s own labs to be “a conflict of interest” and therefore won’t accept those results for claims on our bottle labels or website at this time.
We have no such constraints outside of the United States.
| Microbes | USA | EU | Middle East | China | S. Africa |
|---|---|---|---|---|---|
| Odour causing bacteria | X | X | X | X | X |
| Broad Spectrum bacteria | - | X | X | X | X |
| Viruses | - | X | X | X | X |
| SARS-CoV2 | - | X | X | X | X |
| Fungi (Mould & Mildew) | X | X | X | X | X |
| Algea | X | X | X | X | X |
| Yeast | X | X | X | X | X |
GOLDSHIELD STUDIES

Evaluation of mould and mildew prevention in coastal, post-hurricane Katrina environment.
Results: Hospital wall showed no visible mold or mildew through completion of evaluation – 41 days.
